User Guide
Complete documentation for researchers and administrators
What is the Balloon Analogue Risk Task?
The Balloon Analogue Risk Task (BART) is a computerized behavioral measure designed to assess risk-taking propensity in laboratory and clinical settings. Developed by Lejuez and colleagues in 2002, the BART has become one of the most widely used experimental paradigms for studying decision-making under uncertainty.
Task Overview
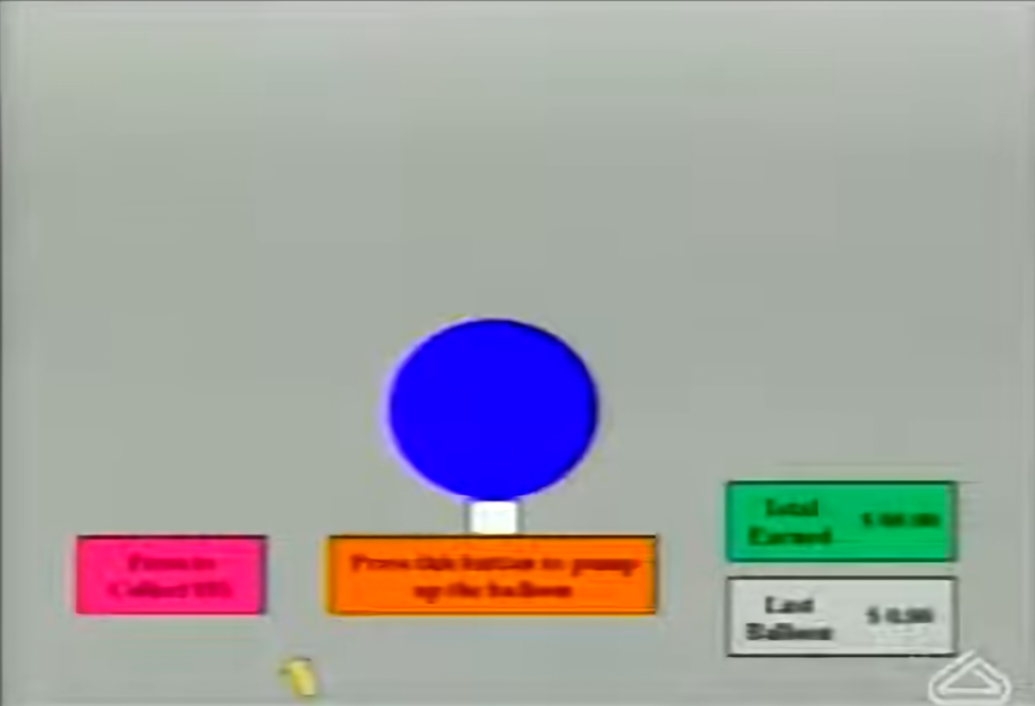
During the BART, participants are presented with a balloon on the screen and have the opportunity to inflate it by pressing a button. Each pump earns a small monetary reward that accumulates in a temporary bank. However, balloons have an unknown explosion point—if the balloon explodes, all money in the temporary bank for that balloon is lost. Participants can choose to "collect" their earnings at any time, transferring the accumulated amount to a permanent bank before the balloon explodes.
What Does It Measure?
The BART provides several key metrics of risk-taking behavior:
- Average Adjusted Pumps: The primary outcome measure, representing the average number of pumps on balloons that did not explode
- Total Pumps: The overall number of pumps across all trials
- Explosion Frequency: How often participants experience balloon explosions
- Total Earnings: The cumulative monetary reward collected
Applications in Research
The BART has been validated across numerous studies and populations, demonstrating significant correlations with real-world risk behaviors. Researchers use the BART to study:
- Substance use and addiction vulnerability
- Gambling behavior and problem gambling
- Health-related risk behaviors
- Decision-making in clinical populations
- Neurobiological correlates of risk-taking
- Developmental changes in risk propensity
Historical Context
The original BART was introduced in a seminal 2002 publication in the Journal of Experimental Psychology: Applied. Since then, it has been cited thousands of times and adapted for various research contexts, including neuroimaging studies, cross-cultural research, and clinical intervention trials. The task's ecological validity—its demonstrated relationship with real-world risk behaviors—has made it particularly valuable for translational research bridging laboratory findings and practical applications.
Note: This platform provides a validated implementation of the BART following the original protocol specifications, ensuring consistency with published research standards.
Getting Started with the BART Platform
This guide will walk you through setting up your research study using our BART platform, from creating an account to distributing experiments to participants.
Creating a Researcher Account
To begin using the platform, you'll need to create a researcher account:
- Navigate to the home page and click "Researcher Portal"
- Select "Create Account" and provide your institutional email address
- Complete the registration form with your information and research credentials
- Verify your email address through the confirmation link sent to your inbox
- Once verified, log in to access your researcher dashboard
Important: We recommend using an institutional email address (.edu, .ac, or .org) to ensure uninterrupted access to your research data.
Creating an Experiment
From your researcher dashboard, you can create customized BART experiments:
- Click "Create New Experiment" in your dashboard
- Provide a descriptive name for your study (e.g., "Adolescent Risk Study - Fall 2024")
- Configure task parameters:
- Number of balloon trials (default: 30)
- Explosion probability range
- Reward values per pump
- Display settings and instructions
- Set data collection preferences and participant information fields
- Generate your unique experiment code (format: ABC-1234)
Browser vs. Desktop Versions
Our platform offers two deployment options to suit different research needs:
Browser Version (Web-Based):
- Accessible from any device with internet connection
- No installation required for participants
- Ideal for remote data collection and online studies
- Real-time data synchronization to secure servers
- Best for: Large-scale surveys, remote participants, multi-site studies
Desktop Application:
- Downloadable application for Windows, Mac, and Linux
- Functions offline without internet connection
- Reduced latency for precise reaction time measurements
- Local data storage with batch upload capability
- Best for: Laboratory settings, controlled environments, neuroimaging studies
Technical Recommendation: For research requiring millisecond-precision timing (e.g., EEG/fMRI studies), we recommend the desktop application to minimize browser-related latency variability.
Distributing Experiments to Participants
Once your experiment is configured, you can distribute it to participants using several methods:
Method 1: Direct Code Distribution
- Share the generated experiment code (e.g., PSY-2401) with participants
- Participants enter the code on the home page under "Participant Access"
- Ideal for in-person laboratory studies
Method 2: Direct Link
- Generate a direct URL link from your dashboard
- Distribute via email, survey platforms, or participant portals
- Link automatically loads the correct experiment configuration
Method 3: QR Code
- Generate a QR code for your experiment
- Display in laboratory settings or on recruitment materials
- Participants scan to immediately access the task
Tips for Successful Data Collection
- Test thoroughly: Run through the complete task yourself before deploying to participants
- Provide clear instructions: Ensure participants understand the task mechanics and objectives
- Consider practice trials: Allow participants to complete 3-5 practice balloons before beginning the recorded experiment
- Monitor progress: Use the dashboard to track completion rates and identify technical issues
- Backup data regularly: Export data frequently throughout collection period
Support: If you encounter any technical difficulties during setup, please contact our research support team through the Contact page.
Content In Development
The comprehensive BART guide is currently being written and will be available soon.
This section will include detailed theoretical background, methodological considerations, scoring guidelines, and interpretation frameworks.
Download PDF Guide (Coming Soon)Terms of Use
The BART Research Platform is provided as a free resource for the scientific community to facilitate behavioral research. By using this platform, you agree to the following terms and conditions:
Academic and Research Use
This platform is intended exclusively for academic research, clinical assessment, and educational purposes. Use for commercial purposes, including but not limited to employee screening, consumer assessment, or market research, is strictly prohibited without prior written authorization.
Citation Requirements
Researchers using this platform are required to provide appropriate citations in all publications, presentations, and reports that include data collected using this system:
For the original BART paradigm, cite:
For this platform/software, cite:
Modifications and Adaptations
If you modify the task parameters or procedures from the standard BART protocol, you must clearly describe these modifications in your methodology section. Substantial deviations from the original paradigm should be noted when interpreting results in the context of existing literature.
Data Privacy and Security
As the researcher, you are responsible for ensuring compliance with all applicable data protection regulations (including GDPR, HIPAA, etc.) in your jurisdiction:
- Obtain appropriate informed consent from all participants
- Do not collect personally identifiable information unless necessary and approved by your IRB
- Ensure secure storage and transmission of participant data
Institutional Review
Researchers must obtain approval from their Institutional Review Board (IRB) or Ethics Committee before collecting data from human participants. This platform does not provide IRB approval or substitute for institutional oversight.
Data Ownership and Responsibility
You retain full ownership of all data collected through this platform. We do not access or analyze your research data.